How is AI Redefining Medicine in the UK?

In an interview with the Wired Magazine in 2024, Emmanuel Macron was highly complimentary of AI in healthcare, describing it as 'personalised and preventative healthcare medicine and treatment'.
France is among a large number of nations which are researching the implementation of AI in medicine – “France 2030” outlines an extra €250 billion for AI in its government healthcare spending plan. Is there scope for the UK in this field?
Biotechnology
The global biotechnology industry was valued at a staggering 1.95 trillion USD, and, according to statistics released in 2024, is expected to grow to 5.90 trillion USD by 2034. At the moment, machine learning algorithms are being used to filter through vast libraries and databases of chemical compounds, predict molecular interactions, and identify the best candidates for drug development. AI is also being used increasingly to find patterns and sequences in genomic analysis, assisting researchers to quickly sort through data.
Firms such as Exscientia (merging with Recursion), headquartered in Oxford, are implementing AI in existing drug discovery platforms. Their approach combines automation, machine learning, and biological data to design candidate drugs more efficiently than traditional pharmaceutical pipelines. Notably, Exscientia produced one of the first AI-designed molecules to enter human clinical trials — a sign that AI drug discovery is moving from theory into practice.
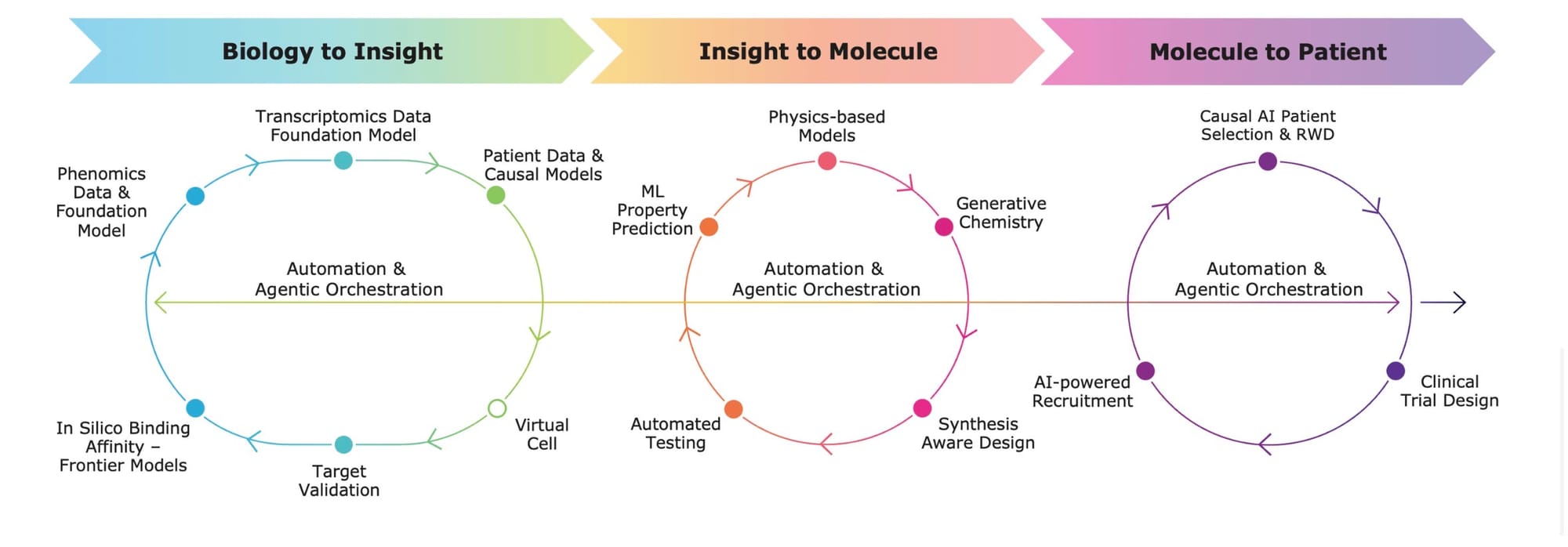
A sample AI driven drug discovery pipeline | Recursion
The NHS and Clinical Implementation
The real test of AI’s value lies in everyday clinical care. Here, the United Kingdom has a unique strategic advantage: the scale and structure of the National Health Service, or as we fondly know it, the NHS.
Since the NHS operates as a largely unified national system, it holds extensive patient datasets — a valuable resource for training medical AI models (as long as the data is used under strict supervision and regulation). Across the country, AI is already being piloted in several clinical environments:
- Radiology: AI tools can flag suspected cancers on X-rays and CT scans, helping radiologists prioritise urgent cases. This is as a result of the AI's training on huge datasets, looking for patterns on both written and graphic formats.
- Ophthalmology: Automated screening for certain coniditons is reducing diagnostic backlogs.
- Pathology: Machine learning assists in identifying abnormal cells in samples.
- Primary care triage: AI chatbots and other tools are helping manage patient flow.
One example is the work of DeepMind Health, part of Google DeepMind, which previously collaborated with NHS trusts on algorithms for detecting acute kidney injury and analysing eye scans. Although some early partnerships raised data-governance concerns, they also demonstrated the clinical potential of well-deployed AI systems.
Benefits:
If implemented wisely, AI could deliver several major advantages to UK healthcare, redefining it in our age:
1. Earlier diagnosis
AI systems are great at pattern recognition. In conditions such as cancer and cardiovascular disease, earlier detection can dramatically improve survival rates. Automated screening may help catch disease before symptoms worsen.
2. Personalised treatment
By integrating genomic and lifestyle data, AI enables more tailored therapies. This may improve treatment success while lowering side effects.
3. Operational efficiency
The NHS faces persistent workforce pressures. AI-assisted triage, scheduling and documentation could free clinicians from routine administrative tasks, allowing more time for direct patient care.
4. Accelerated drug development
AI-driven biotechnology may shorten development timelines and reduce costs, bringing new treatments to patients faster and more affordably.
Challenges and Ethical Considerations
Yet AI in medicine must be deployed safely and ethically, since the system itself would be dealing with confidential patient records.
Data privacy and trust remain paramount. NHS patient data is highly sensitive and public confidence depends on transparency. The UK’s Medicines and Healthcare products Regulatory Agency and the National Institute for Health and Care Excellence are developing frameworks to ensure that AI tools are safe, effective, and ethically deployed.
Bias in algorithms is another potential worry. If training datasets are unrepresentative, AI systems may perform unevenly across different populations and groups, delivering incorrect treatment and potentially widening health inequalities.
The UK’s Strategic Opportunity
Despite global competition, the United Kingdom remains well positioned in the AI-medicine race thanks to:
- World-class universities
- A unified national health system
- An active regulatory environment
Government strategies such as the UK Life Sciences Vision and the AI Roadmap signal continued commitment to this sector. If investment, regulation, and clinical adoption/trials remain steady, the UK could become a leading hub for AI-enabled healthcare innovation.
Artificial intelligence is not about replacing doctors or removing the human touch from medicine. Instead, it is strengthening it. In laboratories across Oxford and hospitals within the NHS, AI is already helping clinicians detect disease earlier and assisting to manage overwhelming workloads. For patients, this could mean faster diagnoses, more personalised care and, ultimately, better outcomes.
However, progress must be made with responsibility. Public trust and data protection cannot be forgotten. If the UK can balance innovation with regulation, AI will modernise healthcare for generations to come.
Bibliography
- Thompson, Nicholas. “Emmanuel Macron Q&A: France’s President Discusses Artificial Intelligence Strategy.” Wired, WIRED, 31 Mar. 2018, www.wired.com/story/emmanuel-macron-talks-to-wired-about-frances-ai-strategy/.
2. “France 2030 | Info.gouv.fr.” Info.gouv.fr, 2025, www.info.gouv.fr/grand-dossier/france-2030.
3. Grand View Research. “Biotechnology Market Growth Analysis Report, 2021-2028.” Www.grandviewresearch.com, Mar. 2021, www.grandviewresearch.com/industry-analysis/biotechnology-market.
- Statifacts. “Biotechnology Market Statistics 2025-2034.” Statifacts, 25 Apr. 2025, www.statifacts.com/outlook/biotechnology-market.
5. Johns. “The Future of AI in the Biotechnology Industry.” Johns Hopkins Advanced Academic Programs, 2026, advanced.jhu.edu/about/on-the-advance/mastering-your-future/the-future-of-ai-in-the-biotechnology-industry/.
6. “Technology.” Recursion.com, 2025, www.recursion.com/platform.
7. Wills, Todd. “AI Drug Discovery: Assessing the First AI-Designed Drug Candidates to Go into Human Clinical Trials.” Cas.org, CAS, 23 Sept. 2022, www.cas.org/resources/cas-insights/ai-drug-discovery-assessing-the-first-ai-designed-drug-candidates-to-go-into-human-clinical-trials?utm_source=chatgpt.com. Accessed 16 May 2026.
- Burki, Talha. “A New Paradigm for Drug Development.” The Lancet Digital Health, vol. 2, no. 5, May 2020, pp. e226–e227, https://doi.org/10.1016/s2589-7500(20)30088-1.
9. “Scaling Streams with Google.” Google DeepMind, 6 May 2025, deepmind.google/health.
10. MHRA. “Medicines and Healthcare Products Regulatory Agency.” GOV.UK, 2025, www.gov.uk/government/organisations/medicines-and-healthcare-products-regulatory-agency.
- NICE. “NICE | the National Institute for Health and Care Excellence.” NICE, NICE, 2025, www.nice.org.uk/.
- Obermeyer, Ziad, et al. “Dissecting Racial Bias in an Algorithm Used to Manage the Health of Populations.” Science, vol. 366, no. 6464, 25 Oct. 2019, pp. 447–453, https://doi.org/10.1126/science.aax2342.



