Lithium-ion Batteries and Sustainable Alternatives

Lithium-ion batteries (LIBs) are an essential component of modern technology, especially in portable electronics (phones, tablets and laptops) and electric vehicles [1] such as cars and scooters (J.B. Dunn et al., 2015). However, the limitations of our dependency on LIBs, such as the scarcity of sources, the fragile geopolitics of the supply chain and potential safety risks, require us to implement a range of different sustainable energy storage technologies alongside LIBs. This article looks at how LIBs can work alongside such emerging chemistries, evaluating their feasibility, which can reduce rare-earth dependency for energy storage and environmental impact [2] (B. Truchot et al., 2025) through a multi-chemistry approach.
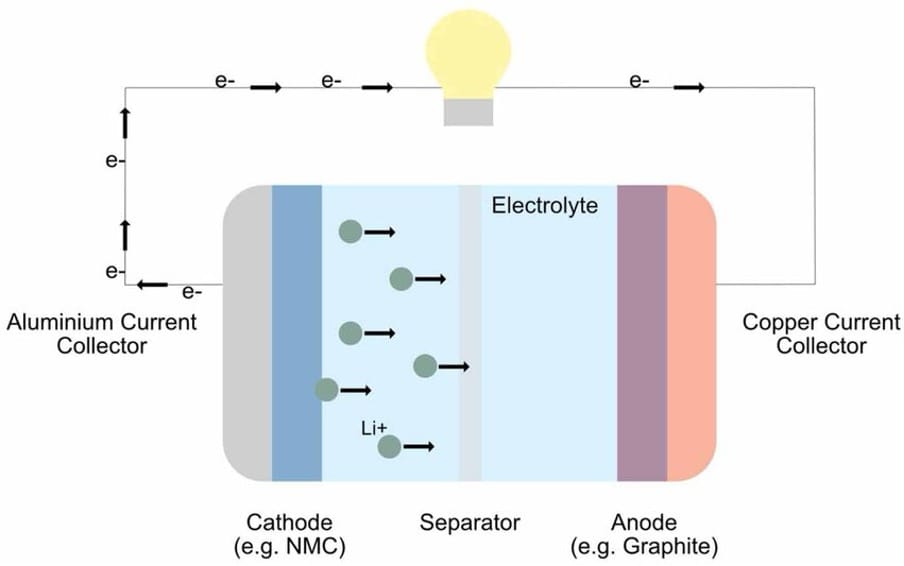
The Chemical Mechanism Behind a Lithium-ion Battery
The traditional mechanism behind a LIB involves a reversible electrochemical process where Li+ ions move between an anode (usually made of graphite) and a cathode (usually a metal oxide) through a liquid organic electrolyte, allowing for fluid movement. The primary conducting salt in commercial LIBs is lithium hexafluorophosphate (LiPF6) [3] (L. Sheng et al., 2023), and during discharge, the ions move from the anode to the cathode, releasing electrons to power the device, while charging reverses this by forcing ions back in between the layers of anode graphite. A thin plastic film acts as a separator that prevents the two electrodes from touching each other.
Industrial manufacturing process of LIBs
During industrial manufacturing, a thick slurry using the solvent N-methyl-2 pyrrolidone (NMP) is spread onto thin metal foils, typically aluminium (cathode) and copper (anode). NMP, being toxic, is evaporated in ovens, and the foils are cut, layered with a separator, then placed into an aluminium or steel casing. With cylindrical cells, the layers are rolled and cased in their shape. Finally, the liquid electrolyte is injected into the cell in a dry environment, as moisture can cause a violent reaction with lithium. An initial charging and discharging forms Solid Electrolyte Interphase (SEI), a protective layer on the anode, preventing the electrolyte from continuously reacting with it, making the Li+ ions stable in the long term.
The advantages of using Li+ ion batteries
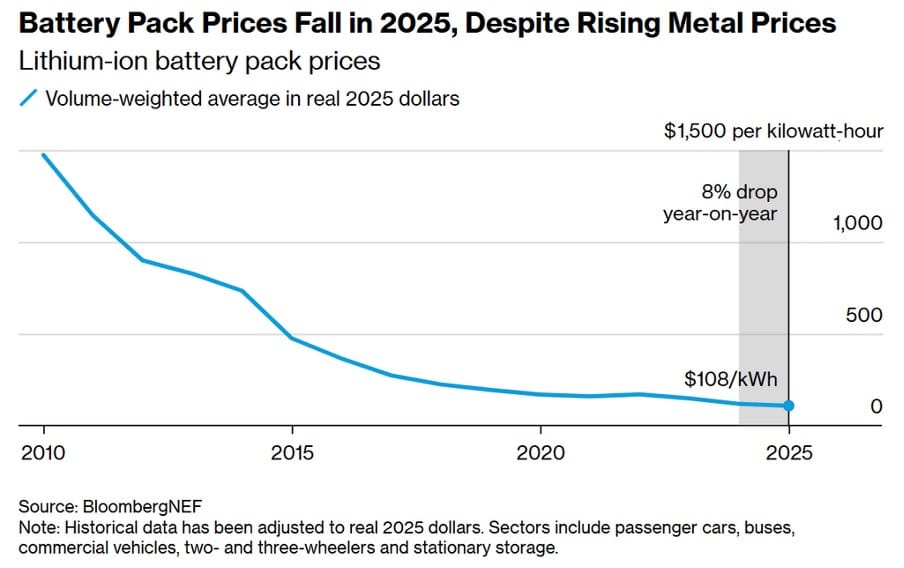
LIBs continue to be the most widely used energy storage medium for a range of quality reasons: they have a very high energy density of 200-300 Wh/kg, which is nearly twice that of other competitors, such as sodium-ion batteries, which have energy densities of 140-160Wh/kg, enabling them to power devices such as electric vehicles for longer periods. Moreover, prices have dramatically dropped over the past 15 years, with decreases of 93% since 2010 (BloombergNEF, 2025). With a record low of $108/kWh at the end of 2025, the majority of manufacturers continue to push for LIBs due to their lower costs of production and higher quality compared to alternatives.
The Non-Sustainability of Use
While LIBs are a key component in the green transition, they also pose several sustainability issues that challenge long-term viability. With high demand in the electronics industry, long-term supply stability is unsustainable. Moreover, the extraction of materials, particularly lithium, is highly water-intensive and can cause toxic runoff and water pollution, and manufacturing also involves hazardous chemicals such as NMP, which pose environmental and safety risks during industrial use. The batteries present significant safety risks as they can cause fires, explosions and the emission of toxic substances when they might be damaged or short-circuited. Environmentally, there is little recycling of the batteries’ end-of life waste, with a mere 5% being recycled [4] (M. Etude et al., 2024), with the total amount estimated by the IEA being expected to reach 8 million tons by 2040 [5] (Z. Baum et al., 2022). With little recycling infrastructure and soaring use of LIBs, the batteries can cause severe damage to the environment, such as the leaching of heavy metals into ecosystems, as well as creating 48% of waste fires in the UK each year.
Sodium-Ion Batteries
With the unsustainable aspects of lithium-ion batteries, it is sensible to turn to more sustainable alternatives. Right now, the most commercially used alternative to LIBs is sodium-ion batteries (SIBs), as sodium is 1200 times more abundant than lithium naturally (R. Clément, 2022), providing a solution to the limited sources of lithium by simply replacing the lithium with sodium in the battery. Due to this abundance, there are large potential for cost reduction for manufacturers, providing a more sustainable alternative. Moreover, SIBs are particularly useful in extreme conditions, as they can maintain 90% efficiency at -40°C (IEA, 2026) while LIBs lose most of their efficiency. SIBs are notably safer than LIBs, as seen in their higher thermal runaway temperatures of 220-260°C compared to 170-220°C in LIBs (ESS, 2026). However, their energy density of 140-160 Wh/kg remains much lower than that of LIBs, requiring larger and heavier battery packs and also limiting their use in electric vehicles. Thus, SIBs would be a sustainable alternative for uses such as stationary grid storage.
Solid-State Batteries
A significant risk in LIBs is that of it catching fire or exploding upon damage, short-circuiting or overheating. By replacing the flammable liquid electrolytes in these batteries with more stable solid materials, solid-state batteries (SSBs) are inherently non-flammable, providing a much safer option. SSBs can operate safely at temperatures of -40 to 120°C [6] (R. Shang et al., 2025), allowing for a much larger range than that of LIBs (-20 to 60°C). Furthermore, SSBs can reach energy densities of up to 500 Wh/kg (CAS, 2026) in comparison to the conventional limit of 250 Wh/kg in LIBs using liquid electrolytes, thus allowing for more compact batteries that can fit into small spaces, such as in portable devices. These might allow electric vehicles to travel distances as much as 600 miles per charge instead of the normal 250-300, with significantly faster charging, and a longer lifespan of 8000-10000 charge cycles (Enertherm, 2025) compared to around 2000 in LIBs. However, SSBs have higher manufacturing costs, with some estimates suggesting it is up to 8 times more expensive per kilowatt-hour than normal LIBs, thus lacking attraction from manufacturers. SSBs offer a safe, high- capacity prospective alternative to liquid electrolyte LIBs, though high costs provide a challenge.
Redox Flow Batteries
Redox Flow Batteries (RFBs) depart from the traditional concept of energy storage. LIBs store energy in two external liquid electrolyte tanks. During charging and discharging, the liquids are pumped into a central stack, separated by a membrane. Reduction and oxidation occur at the membrane surface, allowing electron flow to generate electricity without having to degrade physical electrodes. In this process, energy capacity can be easily increased by making the tanks larger. RFBs are advantageous in that they have longer cycle life (1000 compared to 500 in LIBs [7] (K. Vafaeva and P. Sanjeeva, 2024)) and have lower environmental degradation and production of toxic materials. As with many alternatives to LIBs, they lack high energy density; RFBs only have one of around 100 Wh/kg, thus excluding use in portable electronics, though they can act as a primary alternative to sustainable grid-based storage. Being liquid-based, RFBs eliminate the thermal runaway hazards in liquid electrolyte LIBs, creating a safe, scalable alternative.
Iron Phosphate Cathodes
Traditionally, in LIBs, Nickel Manganese Cobalt (NMC) cathodes are used (in 2022, the global market share was 60% compared to 30% lithium iron phosphate (LFP), the second most used battery chemistry (IEA, 2023)). However, in the past few years, LFP batteries have risen to over 40%, driven by Chinese demand. Popularity in LFPs as opposed to NMCs is driven by the 40% cheaper manufacturing cost (IEA, 2026) as LFPs use commonly available iron and phosphate, while NMCs must source scarce, expensive nickel and cobalt resources. For this reason, iron phosphate cathodes are more sustainable by using abundant commodities, while also having a higher thermal runaway threshold of 270°C compared to 210°C in NMCs. With end-of-life waste, nickel and cobalt are heavy metals and carcinogens, while iron and phosphate are ecologically safe, thus reducing environmental contamination caused by landfills. Within LIBs themselves, LFPs, therefore, act as a sustainable alternative to the traditional NMCs, reducing the use of heavy metals.
Summary and Remarks
LIBs are a key component in transitioning to a green future, though they themselves face sustainability challenges. With issues such as thermal runaway, fire risks, the use of hazardous materials and very little end-of-life recycling, LIBs face many sustainable alternatives, such as solid-state systems, redox flow batteries, sodium ion batteries and lithium iron phosphate batteries that can aim to reduce as many of these problems as possible. However, LIBs are prized for their high energy density, something which other alternatives cannot easily match. With ever-increasing demand for batteries in electronics and vehicles, these sustainable chemistries must be used maximally with LIBs to meet our storage needs with a reliable, circular battery ecosystem for the future.
Bibliography
- Dunn, Jennifer B., et al. "The significance of Li-ion batteries in electric vehicle life-cycle energy and emissions and recycling's role in its reduction." Energy & Environmental Science 8.1 (2015): 158-168. https://pubs.rsc.org/en/content/articlelanding/2015/ee/c4ee03029j
- Truchot, Benjamin, et al. "Focus on the Environmental Impact of Lithium-Ion Batteries." Chemical Engineering Transactions 116 (2025): 241-246. https://www.cetjournal.it/index.php/cet/article/view/CET25116041
- Sheng, Li, et al. "Unravelling the hydrolysis mechanism of LiPF6 in electrolyte of lithium ion batteries." Nano Letters 24.2 (2023): 533-540.https://pubs.acs.org/doi/10.1021/acs.nanolett.3c01682
- Etude, Martin C., et al. "Recycling lithium-ion batteries: A review of current status and future directions." Sustainable Chemistry One World 4 (2024): 100027. https://www.sciencedirect.com/science/article/abs/pii/S2950357424000271
- Baum, Zachary J., et al. "Lithium-ion battery recycling─ overview of techniques and trends." (2022): 712-719.https://pubs.acs.org/doi/full/10.1021/acsenergylett.1c02602
- Shang, Ruoxu, et al. "A comprehensive review of solid-state lithium batteries: Fast Charging characteristics and in-operando diagnostics." Nano Energy 142 (2025): 111232.https://www.sciencedirect.com/science/article/pii/S2211285525005919
- Vafaeva, Khristina Maksudovna, and P. Sanjeeva. "Comparative analysis of lithium-ion and flow batteries for advanced energy storage technologies." MATEC Web of Conferences. Vol. 392. EDP Sciences, 2024.https://www.matecconferences.org/articles/matecconf/abs/2024/04/matecconf_icmed2024_01176/matecconf_icmed2024_01176.html

![Image via [Adobe Stock]](/content/images/size/w600/2026/03/IMG_0874.jpeg)
